Dermal/Skin Models

Imiquimod (IMQ) Induced Psoriasis Model
Psoriasis is a common skin disorder characterized by focal formation of inflamed, raised plaques that shed scales from excessive growth of epithelial cells and involves the following histological changes in the skin:
- hyperplasia of epidermal keratinocytes
- vascular hyperplasia and ectasia
- infiltration of T lymphocytes, neutrophils and other types of leukocytes in the affected skin.
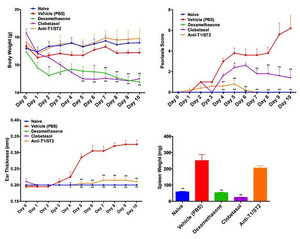
As experts in skin inflammation modeling, MLM Medical Labs is proud to present its extensive experience as one of the first laboratories to have validated the imiquimod induced psoriasis model in mice. This rapid model of psoriasis-like inflammation involves the IL-17/IL-23 axis. Imiquimod is a potent TLR7/8 ligand and potent immune modulator. Application of imiquimod on the back and ear of mice induces a psoriasis-like inflammation.
Assessments:
- Clinical Score
- Ear Thickness
- Biomarker analysis (protein or mRNA)
- Cell Populations in Spleen/Lymph Node by FACS
- Histology/IHC
- PK blood collections
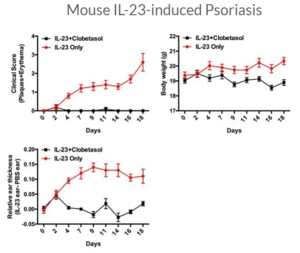
IL-23-Induced Psoriasis Model
Species available: Mouse IL-23 Psoriasis, Rat IL-23 Psoriasis
Recent research in psoriasis has been focused on the IL-23/Th17 pathway. IL-23 stimulates and promotes differentiation of Th17 cells. IL-23 is a heterodimeric cytokine with two subunits. It drives the Th17 response by its binding and signaling through its receptor subunits. When the IL-23R is activated, it promotes the development of Th17 cells and the resulting production of cytokines such as IL-17A, IL-17F, and IL-22 - all which are involved in mediating psoriasiform changes.
The IL-23 induced psoriasis model involves the injection of IL-23 into the ear of C57Bl/6 and is dependent on IL-22 for the development of dermal inflammation and acanthosis. IL-22 induces keratinocyte proliferation and epidermal hyperplasia contributing to epidermis thickening.
Assessments:
- Clinical Score
- Body weight
- Ear Thickness
- Biomarker analysis (protein or mRNA)
- Cell Populations in Spleen/Lymph Node by FACS
- Histology/IHC
- PK blood collections
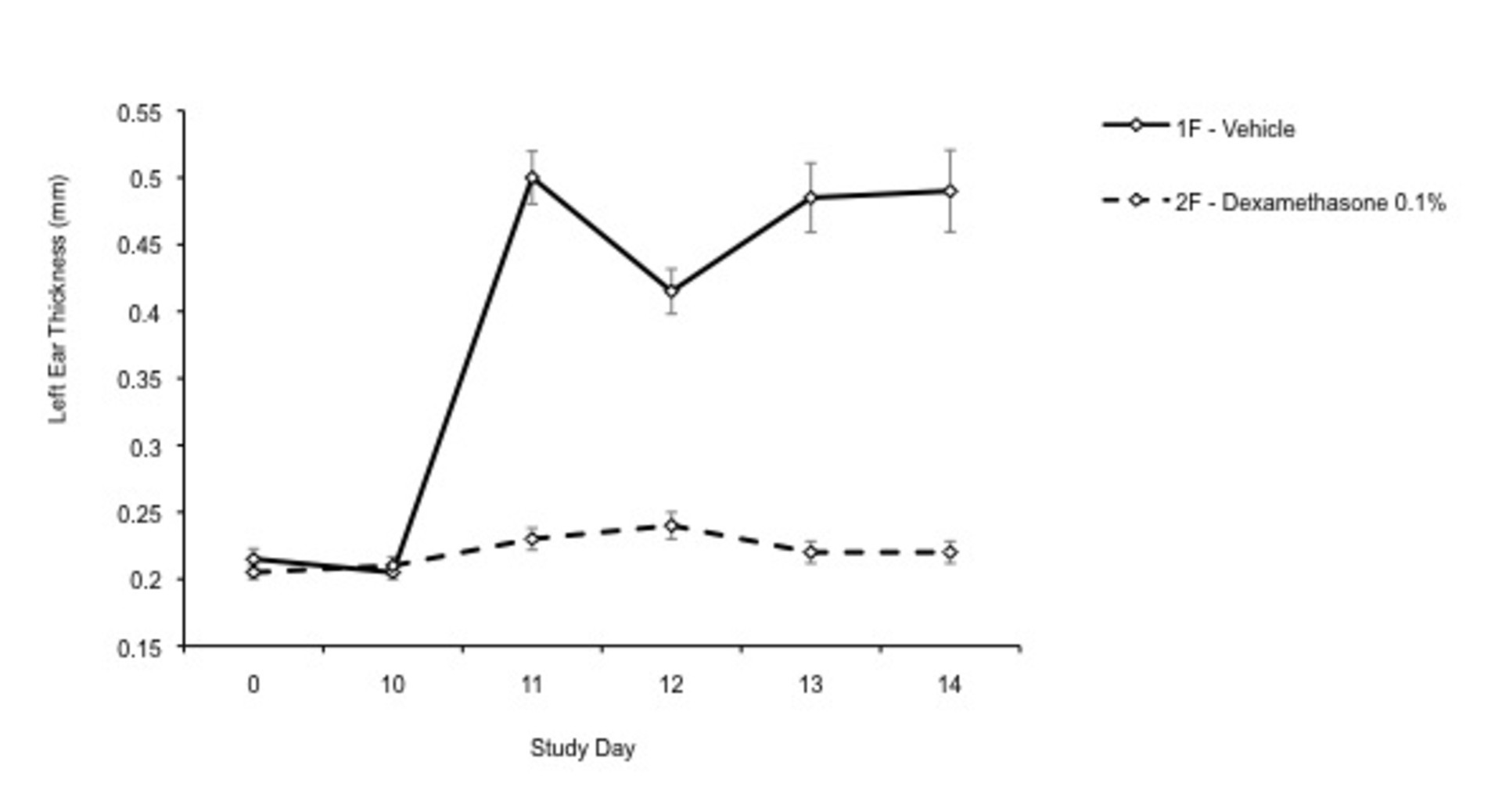
FITC & DNCB-Induced Contact Dermatitis Model
Species available: Mouse FITC Dermatitis, Mouse DNCB Dermatitis
Allergic contact dermatitis, which is also known as contact hypersensitivity, indicates an overreaction of the body's immune system to a normally harmless substance. Irritant contact dermatitis may occur on first exposure to the substance, while contact hypersensitivity requires that a previous sensitization to the substance has occurred. Unlike atopic eczema, in contact dermatitis this sensitization almost exclusively occurs via the skin. Clinically, allergic contact dermatitis is a common cause of occupational allergic dermatitis and may be caused by a wide variety of substances such as heavy metals (i.e. nickel), a variety of petrochemicals (many of which are used in dyes and paints) or plant-derived compounds. The rash associated with poison oak/ivy exposure is also an example of allergic contact hypersensitivity (urushiol-induced contact dermatitis).
In contact hypersensitivity, the sensitizing agent is usually a small molecule which alone would be insufficient to generate an immune response. However, during the sensitization phase of allergic contact dermatitis, these small molecules become highly immunogenic by binding to proteins within the skin, effectively acting as a hapten. These proteins are then picked up by Langerhans cells of the skin which subsequently present haptenated peptides to T cells in the draining lymph nodes. Re-exposure to the sensitizing agent following sensitzation results in expansion of these T cells and infiltration into the skin where they induce the recruitment of additional cells and ultimately resulting in the dermatitis.
About FITC & DNCB-induced Contact Dermatitis Models at MLM:
During the sensitization phase skin on the flanks is exposed topically to either FITC or DNCB on two occasions. During this phase of the models these small molecules become immunogenic by binding to proteins within the skin and act as haptens. These haptenated proteins are then picked up by Langerhans cells of the skin and subsequently present haptenated peptides to T cells in the draining lymph nodes. In the challenge phase, mice are then challenged on the ear with the FITC or DNCB. During this phase haptenated peptides are again presented to T cells in the draining lymph nodes, however as the cells are already primed an inflammatory response occurs leading to the development of dermatitis. In the DNCB induced model the inflammation is Th1 mediated (IFN-g, IL-12) also with a role for CD8+ T cells, and macrophage like monocytes. In the FITC induced model is Th2 (IL-4, IL-5) mediated, a role for eosinophils and mast cells has also been demonstrated in this model.
These models share many of the features of human contact dermatitis, both sensitization and challenge occurs epicutaneously, during the sensitization phase no pathology is observed within exposed skin but on subsequent exposure exposed skin becomes red, swollen and hyperplasic, there is also a loss of barrier function in the skin. The distinct sensitization and challenge phases and the distinct locations of these events allow the modulation of the immune response at either stage.
Assessments:
- Ear thickness
- Circulating or tissue level Biomarker analysis (protein or mRNA)
- Cell Populations in Spleen/Lymph Node by FACS
- Histology/IHC
- PK blood collections
Ear Thickness in the FITC Contact Dermatitis Study
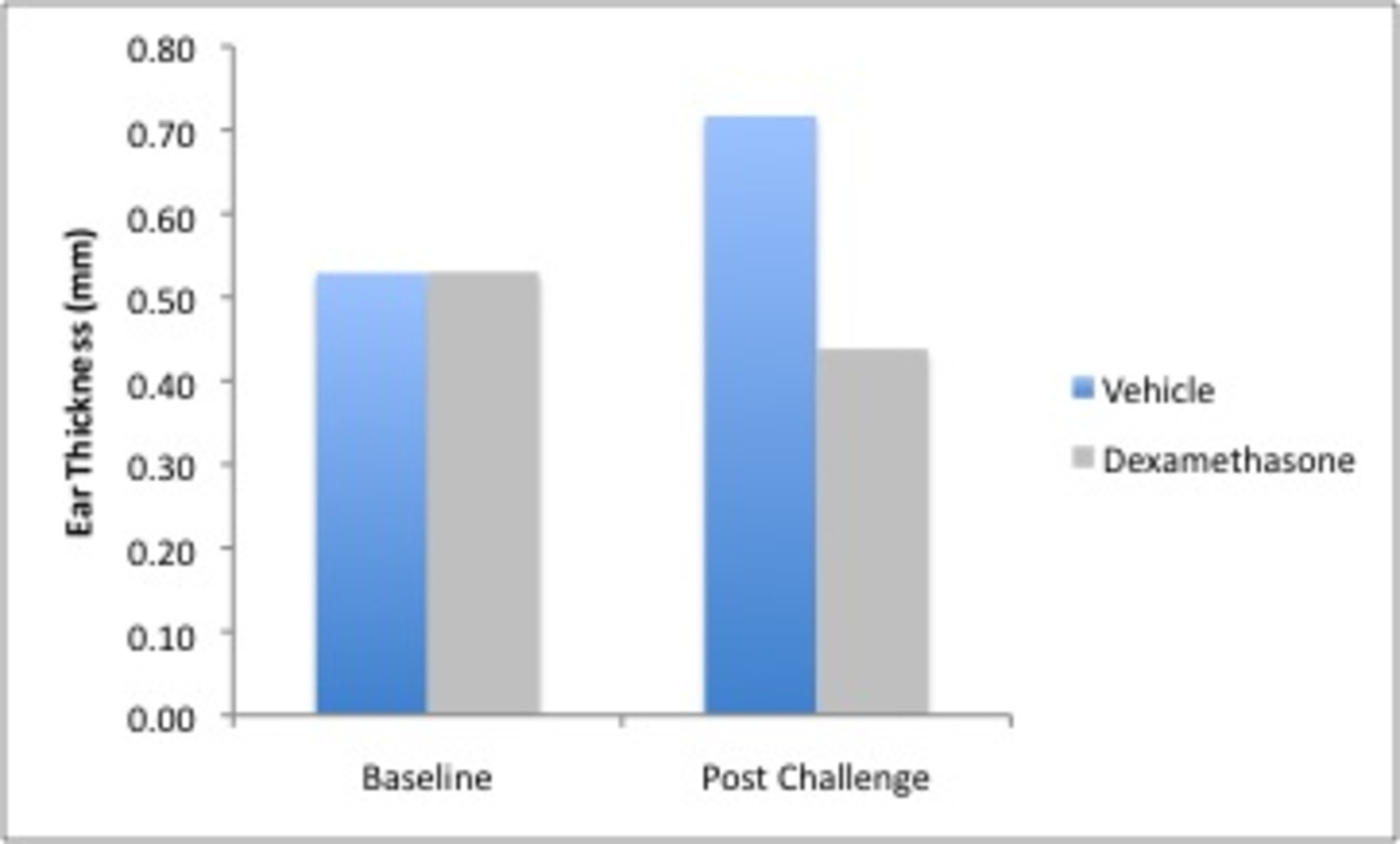
Oxazalone-Induced DTH Model
Species available: rat DTH
Our Delayed Type Hypersensitivity (DTH) model evaluates the cell-mediated immune response to repeated exposure of skin to oxazolone. This results in a DTH reaction involving CD4+, CD8+ T cells and macrophages, resulting in local skin inflammation characterized by an increased thickness of the skin. An initial sensitization is followed by a challenge, which sets off a release of cytokines initiating a robust inflammatory response. Below are the assessment types and study data.
Assessments:
- Ear thickness
- Circulating or tissue level Biomarker analysis (protein or mRNA)
- Cell Populations in Spleen/Lymph Node by FACS
- Histology/IHC
- PK blood collections
Ear Thickness in the DTH Study
Passive cutaneous Anaphylaxis Model
Species available: Mouse PCA, Rat PCA
Passive Cutaneous Anaphylaxis (PCA) is an immediate dermal response to an allergan-IgE interaction. It is based on a passively transferred anaphylactic reaction from antiserum. Subjects are sensitized with antiserum and following a latent period, subjects are injected IV with Evans blue dye bound to the antigen. Reaction of the antigen with skin-fixed antibody causes the release of histamine, which increases vascular permeability, permits leakage of the albumin -bound dye, and produces a blue spot at the site of the intradermal injection.
Assessments:
- Dye extravasation (mm)
- Absorbancy of Evans Blue
- Biomarker analysis (protein or mRNA)
- PK blood collections