Study preparation
Diligent but rapid service

Before your first sample is analysed at MLM, our team will already have worked intensively to properly set up your clinical study. Our preparatory services include:
- Preparing a user-friendly lab manual for the clinical trial sites with all the essential and necessary information for a proper sample draw, on site processing, storage and shipment
- Provision of certificates, CVs of key study personnel and reference ranges
- Designing study-specific requisition forms
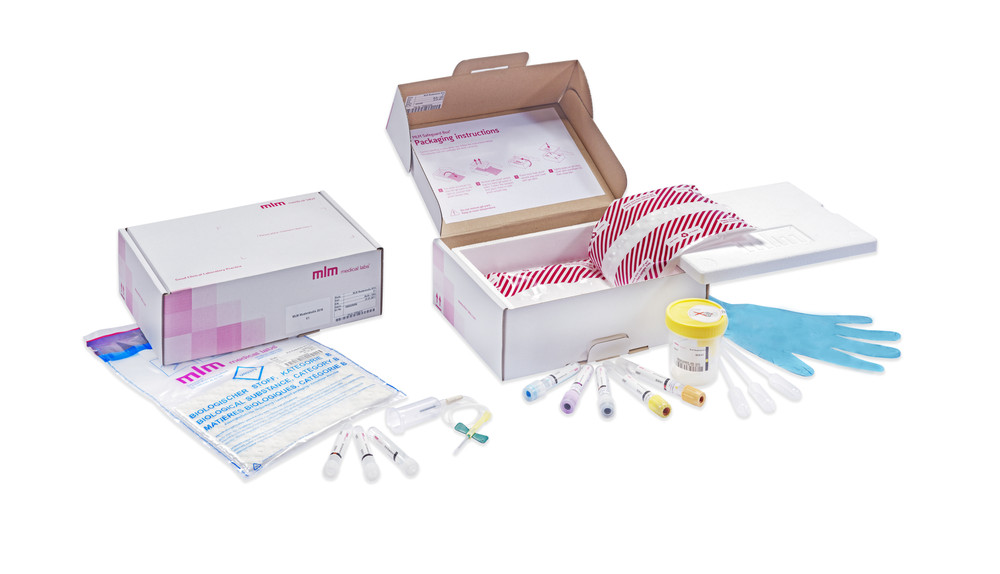
- Designing study-specific sampling kits
- Manufacturing kits through MLM Kit BuildingTM
- Preparing a dedicated database in our LIMS, accessible via mlm onlineTM
- Setting up the study communication plan
- Selecting the most efficient logistics solutions
For EU countries, we require 3 to 6 weeks* from project approval for sampling kit and lab manual delivery. For countries outside the EU, import and customs clearance must be addressed, which may add to the delivery time.
*Depending on the size of the clinical study